 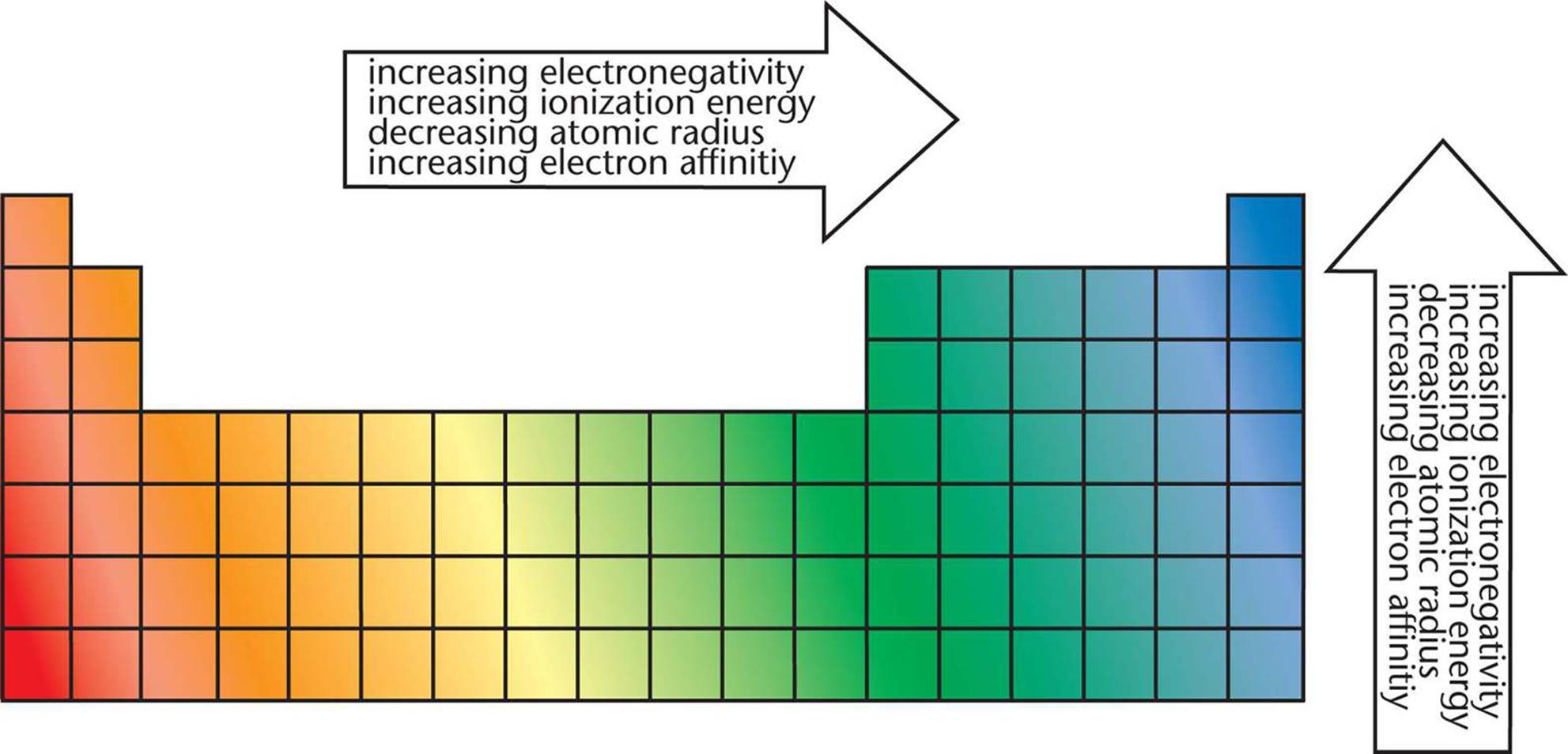
Plan to play the song at least twice to get your students thinking about the table and its general set up. A new version of the song is at Periodic Table Song. In the first part of this activity, students will get to know the periodic table through “The Periodic Table Song,” various versions of which are available on YouTube. Handouts showing the periodic table or a large periodic table wall chartĬlassroom computer projector to show video clips demonstrating chemical reactivity A second set of videos focused on reactivity will encourage students to use their observations to identify trends and predict behavior in reactivity among metals and nonmetals. Purpose: After watching a video that introduces them to the periodic table, students will answer questions that prompt them to identify general patterns in the table. (Physical_and_Theoretical_Chemistry)/Physical_Properties_of_Matter/Atomic_and_Molecular_Properties/Ionization_Energy. *We hope you enjoyed studying this lesson and learned something cool about the Trends in the first ionization energy! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our App to experience our fun, VR classrooms – we promise, it makes studying much more fun! □ * SOURCES: In general, ionization energy rises over a period and falls as a group descends. What is the group trend in the first ionization energies? The first ionization energy decreases as you go to the bottom of a group.Ĥ. What is the trend in 1st ionization energy from top to bottom down a group? 
The general trend of ionization energy in a periodic table is that the energy increases as you move in the direction of hydrogen to helium and decreases as you move from hydrogen to cesium.ģ. The first ionization energy increases as you move from left to right over time, but it decreases as you move down a group.Ģ. How is the first ionization energy trend? Across the periodic table, ionization energy increases from left to right, and as we progress downwards in a group, the ionization energy decreases.ġ.The ionization energy of electrons in different atomic or molecular orbitals is variable.The ease with which electrons can be withdrawn from an atom affects the atom’s reactivity.The energy required to remove an electron from a certain atom is known as ionization energy.Ionization energy increases from Hydrogen to Helium in a period but decreases from Hydrogen to Francium in a group. They are less attracted to the positive charge of the nucleus. The ionization energy is likely to reduce as one proceeds downwards the periodic table because the electrons in the outermost shell are further away from the nucleus and more insulated. As the atomic size reduces, it becomes more difficult to eliminate an electron nearer to a more central nucleus. The low one’s elements tend to be reducing agents.Īs you move across a period, the first ionization energy increases. The ionization energy of an element can be used to determine its reactivity. The more ionization energy there is, the harder it is to remove an electron. It measures an atom’s or ion’s tendency to relinquish an electron or the intensity of the electron binding. The xth ionization energy is the energy needed to eliminate the nth electron after the first x-1 electrons have been withdrawn. The ionization energy of electrons in different atomic or molecular orbitals is variable. atomic number for the major group elements on a graph. We may see the following trend if we describe the trends in first ionization energy vs. As more electrons add around a nucleus, the inner shell electrons shield the outer electrons from the nucleus. Any atom’s outermost valence electrons will have lower ionization energies than the inner-shell electrons. The table below shows some of the ionization energies linked with several elements. It is expressed in kJ/mol, an energy unit similar to calories. The energy required to remove an electron from a certain atom is ionization energy. The ease with which electrons can be withdrawn from an atom affects the atom’s reactivity, and this quantity can be measured and used to make accurate predictions about atom behavior. Similarly, the electrons and protons in an atom are always in conflict. Now, a student’s choice to act individually or with the class depends on the balance. The worst is that a student may be scared for some reason and may run off screaming. A student may carelessly wander off to other strong, attractive forces of a bright crayon, a fluffy toy, or their parents. We all know how difficult it is to teach in a preschool, don’t we? You want the excited young students to stay together and maintain uniformity, but this stability can barely be achieved. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
